|
|
Original Article
| ||||||
| Chemical composition, antioxidant and larvicidal activity of Alchornea laxiflora (Benth) leaf extracts | ||||||
| Frank N. I. Morah1, Dickson N. Uduagwu2 | ||||||
|
1PhD, Professor of Natural Products Chemistry, Chemistry Department, University of Calabar, P.M.B, Calabar, Cross River State, Nigeria
2MSc, Scholar, Chemistry Department, University of Calabar, PMB, Calabar, Cross River State, Nigeria | ||||||
| ||||||
|
[HTML Abstract]
[PDF Full Text]
[Print This Article]
[Similar article in Pumed] [Similar article in Google Scholar] |
| How to cite this article |
| Morah FNI, Uduagwu DN. Chemical composition, antioxidant and larvicidal activity of Alchornea laxiflora (Benth) leaf extracts. Edorium J Pharmacol 2017;1:1–8. |
|
ABSTRACT
|
|
Aims:
To determine the chemical constituents, antioxidant activity and insecticidal potential of Alchornea laxiflora leaf extracts.
| |
| Keywords: Alchornea laxiflora, Antioxidant activity, Anopheles mosquito, Chemical composition, Larvicidal activity | |
|
INTRODUCTION
| ||||||
|
Alchornea laxiflora (Benth.) of the family Euphorbiaceae is a deciduous shrub or small tree and an African medicinal plant. Decoction of the leaf is used for control of inflammations, malaria, bacterial and fungal infections [1]. It is taken orally for the treatment of postpartum pains in Cameroun [2]. Alchornea laxiflora is known to contain a number of flavonoids which include quercetin, quercetin-3-O-b-D-glycopyranoside [3], laxiflorin glycoside [4] and alchornealaxine [5]. The use of natural products or medicinal plants and natural insecticides is becoming very popular because of development of resistance to the conventional synthetic drugs and insecticides. The natural products have the added advantage of being cheap, readily affordable and have reduced risk of side effects. They are also environmentally friendly [6]. Alchornea laxiflora leaf is employed in traditional medical practice for the management of different human health problems. The aim of the present investigation is to identify its chemical constituents which are believed to be responsible for its pharmacological activity. It is also aimed at evaluation of its antioxidant and larvicidal properties. | ||||||
|
MATERIALS AND METHODS
| ||||||
|
Alchornea laxiflora leaves were harvested in the month of July from Ikwo in Eboyi State of Nigeria. It was authenticated by Frank Adeoye of the Herbarium unit, Botany Department, University of Calabar, Nigeria. The leaves were rinsed with distilled water, air dried and ground with a mill. The ground leaf was extracted with petroleum spirit (60–800C) in a Soxhlet extractor and distilled down to give the petroleum ether extract. The leaf residue left after the petroleum ether extraction was re-extracted with ethanol to give the ethanol extract. The petroleum ether extract was fractionated over a silica gel column to give fractions A and B. The gas chromatography-mass spectrometric analysis of these extracts was carried out with GC-MS instrument. The column consists of a 25 m x 0.23 mm fused silica capillary, coated with polydimethylsiloxane (DBP-1) of 1.5 µm film thickness filled in the GC-MS system. The column and oven temperatures were programmed to start from 60°C increasing up to 200°C at 3°C/min. The carrier gas used was helium and at a constant flow rate of 1 ml/min. The antioxidant activity was determined by DPPH free-radical scavenging assay methods. The reduction capacity of the DDPH (2, 2-diphenyl-1-picrylhydrazyl) radical was determined by the decrease in absorbance induced by antioxidants according to the method of Brands-William et al. [7] with a few modifications. Solutions containing 40, 80, 100, 150 and 200 mgdm-3 of both petroleum ether fractions and ethanol extract were separately prepared. 0.1 cm3 of each of these and the standard (containing no extract) were each separately added to 2.9 cm3 of 0.5 mM of DPPH in dimethyl sulfoxide, DMSO. Each mixture was shaken vigorously and left at room temperature for 30 min. The absorbance was taken at λmax 517 nm against a blank. The ability to scavenge the DPPH radical was calculated from this using the following formula: DPPH scavenging effect (%) = A0 - A1/A0 x 100 Anopheles mosquito larvae were used for the larvicidal activity. Twenty-five, third and fourth instar larvae were kept in each of the different 500 cm3 beakers, each containing 10 cm3 of tap water. 1 cm3 of each of the different concentrations of extract was added to the different beakers with exception of the control. Larval mortality was assessed at 24 h intervals for 96 h after which the percentage mortality and probit mortality were calculated. | ||||||
|
RESULTS | ||||||
|
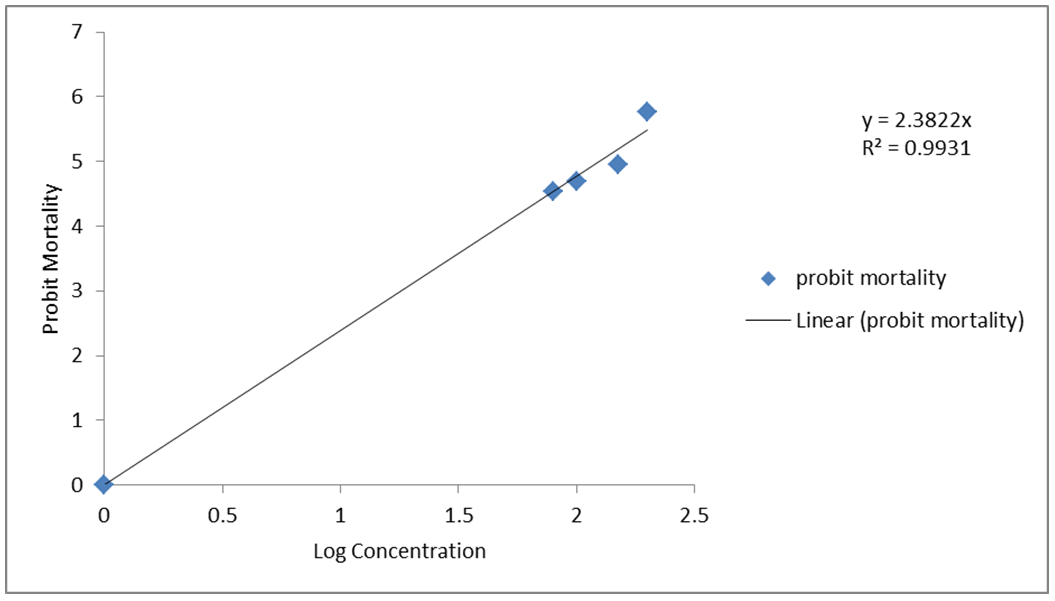
Table 1 and Table 2 give the chemical composition of fractions A and B of the petroleum ether extract of Alchornea laxiflora leaf respectively. Fraction A contains seven identified compounds while fraction B contains twenty-three compounds. With exception of 3-ethyl-5-(2-ethylbutyl)-octadecane which is a hydrocarbon, the rest are oxygenated compounds. Table 3 gives that the ethanol extract of this plant contains seven oxygenated natural products. The antioxidant activities of the ethanol and petroleum ether extracts of Alchornea laxiflora are given in Table 4 and Table 5 respectively. Table 6 gives the antioxidant activity of the ascorbic acid standard. Both extracts possess antioxidant activity which is dose dependent. Larvicidal activity of the leaf extracts against Anopheles mosquito larvae is shown in Figure 1 and Figure 2. The extracts have strong larvicidal activity against Anopheles mosquito larvae (Table 7). | ||||||
| ||||||
| ||||||
| ||||||
| ||||||
| ||||||
| ||||||
| ||||||
| ||||||
| ||||||
|
DISCUSSION
| ||||||
|
Thirty compounds were identified in the petroleum ether extract while seven compounds were identified in the ethanol extract of Alchornea laxiflora leaf. To the best of our knowledge, there is no earlier report of the occurrence of any of these compounds in Alchornea laxiflora. These include astaxanthin, 3-acetoxy-7,8-epoxylanostan-11-o1,4,10,14-trimethylpentadecan-3-one, lycoxanthin, methyl hexadecanoate, rhodopin, linoleic acid, ethyl iso-allocholate, betulin and diisooctyl phthalate. Compounds identified in the ethanol extract include 2, 4-bis (1, 1-dimethylethyl) phenol, ethyl linoleate, eicosyl oleate and glycocholic acid. On the whole thirty-seven compounds were identified from the leaf extracts. This is the first report on the occurrence of these compounds in Alchornea laxiflora. Most of these compounds have pronounced biological activities which include antimicrobial, antiprotozoal, anticancer, antioxidant and anti-arthritic activities [8][9][10][11][12]. Tables 4 Tables 5 Tables 6 show that both ethanol and petroleum ether extracts have high antioxidant activities which is concentration dependent as it increases with increases in concentration of the extracts. They have higher antioxidant activity than the ascorbic acid standard. The petroleum ether extract has higher antioxidant activity than the ethanol extract. The observed antioxidant activities are higher than what was reported for 50% aqueous alcohol extract of this plant [4]. This is attributable to the greater amount of antioxidant potential compounds present in the petroleum ether extract followed by the ethanol extract. The identified antioxidants include 3-acetoxy-7, 8-epoxylanostan-11-one, rhodopin, ethyl iso-allocholate, hexadecanoic acid, 9-octadecenyl hexanoate, eicosyl oleate and astaxanthin [8][9][10][11][12]. This explains the use of Alchornea laxiflora in traditional medicine to control oxidative stress [13]. Table 7 and Figure 1 and Figure 2 show that both ethanol and petroleum ether extracts have larvicidal activity against Anopheles mosquito larva. Since the control showed no larvicidal activity, it is conceivable that the natural products in the Alchornea laxiflora leaf extracts are solely responsible for the observed larvicidal activity. This activity is concentration dependent as an increase in concentration of the extract resulted in higher percentage mortality and hence higher probit mortality of the Anopheles mosquito larvae. The petroleum ether extract generally showed higher larvicidal activity. Some of the identified compounds including methyl hexadecanoate and eicosyl oleate are known to have insecticidal activity [8][10]. Hexadecanoic acid esters and di-n-octylphthalate identified in this plant species have been shown to have larvicidal activity against mosquito larvae [14][15]. The natural substances from plants are more effective and more environmentally friendly than the synthetic commercial insecticides. Natural insecticides are, therefore, better alternatives to the conventional insecticides. | ||||||
|
CONCLUSION
| ||||||
|
The leaf extracts of Alchornea laxiflora have high antioxidant and larvicidal activities. The present study has identified thirty-seven natural products from the plant. These compounds are being reported for the first time in Alchornea laxiflora. Many of the compounds are known to have bioactive activities and are responsible for the pharmacological activities of this plant. | ||||||
|
REFERENCES
| ||||||
| ||||||
|
[HTML Abstract]
[PDF Full Text]
|
|
Author Contributions
Frank N. I. Morah – Substantial contribution to the conception and design, Acquisition of data, Analysis and interpretation of data, Drafting of article, Revising it critically for important intellectual content, Final approval of the version to be published Dickson N. Uduagwu – Acquisition of data Analysis and interpretation of data, Revising the article critically for important intellectual content, Final approval of the version to be published |
|
Guarantor of submission
The corresponding author is the guarantor of submission. |
|
Source of support
None |
|
Conflict of interest
Authors declare no conflict of interest. |
|
Copyright
© 2017 Frank N. I. Morah et al. This article is distributed under the terms of Creative Commons Attribution License which permits unrestricted use, distribution and reproduction in any medium provided the original author(s) and original publisher are properly credited. Please see the copyright policy on the journal website for more information. |
|
|